Each such orbital can be occupied by a maximum of two electrons, each with its own projection of spin m s. As an alternative to the magnetic quantum number, the orbitals are often labeled by the associated harmonic polynomials (e.g., xy, x 2 − y 2). Įach orbital in an atom is characterized by a set of values of the three quantum numbers n, ℓ, and m l, which respectively correspond to the electron's energy, its angular momentum, and an angular momentum vector component ( magnetic quantum number). The term atomic orbital may also refer to the physical region or space where the electron can be calculated to be present, as predicted by the particular mathematical form of the orbital.

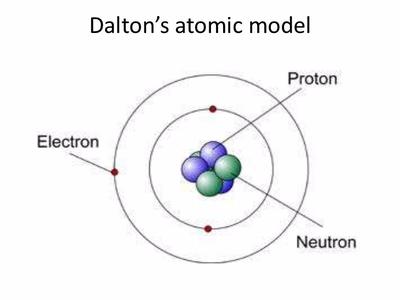
This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. In quantum mechanics, an atomic orbital ( / ˈ ɔːr b ɪ t ə l/) is a function describing the location and wave-like behavior of an electron in an atom. The atomic nucleus is the small, dense region consisting of protons and neutrons at the center of an atom, discovered in 1911 by Ernest Rutherford based on the 1909 GeigerMarsden gold foil experiment. For a more in-depth discussion of the history of atomic models, see atom: development of atomic theory. First established by the French chemist Joseph Proust in 1797, this law states that if a compound is broken down into its constituent chemical elements, then the masses of the constituents will always have the same proportions by weight, regardless of the quantity or source of the original substance. Atomic models have gone through many changes over time, evolving as necessary to fit experimental data. To see the elongated shape of ψ( x, y, z) 2 functions that show probability density more directly, see pictures of d-orbitals below. Atomic model, in physics, a model used to describe the structure and makeup of an atom. Leucippus of Miletus (5th century bce) is thought to have originated the atomic philosophy. Each picture is domain coloring of a ψ( x, y, z) function which depends on the coordinates of one electron. Rutherford model, description of the structure of atoms proposed (1911) by the New Zealand-born physicist Ernest Rutherford. The atomic philosophy of the early Greeks. Previously, we defined an atom as the smallest part of an element that maintains the identity of that element. 1 ), is a fundamental concept that states that all elements are composed of atoms. His work on the first periodic table provided. The modern atomic theory, proposed about 1803 by the English chemist John Dalton (Figure 4.2.1 4.2. The two colors show the phase or sign of the wave function in each region. Antoine Lavoisiers discovery that during chemical change mass is conserved defined the law of conservation of mass and contributed to atomic theory.
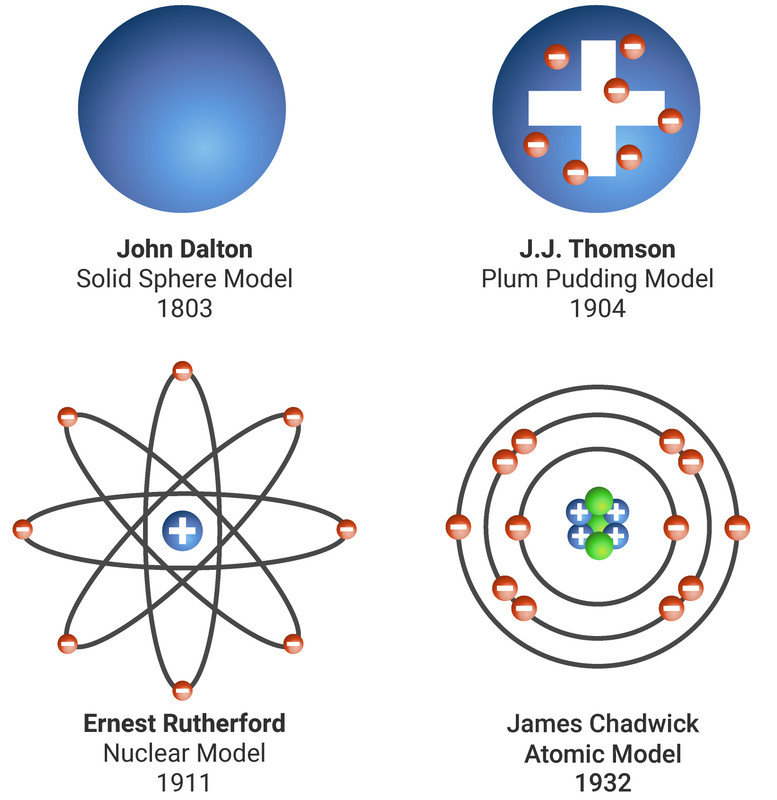
Before Thomsons day, many physicists felt that the field was declining. The shapes of the first five atomic orbitals are: 1s, 2s, 2p x, 2p y, and 2p z. Thomson make his discovery and change atomic theory His breakthrough came in 1897. For the collection of spaceflight orbits, see Orbital shell (spaceflight).
